

Here is a little bit more detail as to what I put in my thesis - click on the thumbnails for associated illustrations. [Note: some of the pictures, particularly maps, are far larger than the screen, so you might have to download and then resize them]
Understanding of the kinetics of metamorphic processes has lagged behind the advances made in the subject through equilibrium thermodynamics, the main stumbling block being an inadequate knowledge of the nucleation process.
For a new mineral to grow as a result of a
prograde metamorphic reaction, a number of processes must operate in
concert. First of all, nuclei of the new mineral
must appear. New material must be added to the embryo crystal coupled
with dissolution of reactant phases, both processes occurring by means
of reactions on the mineral
surfaces. These chemical species must be transported through the
intergranular medium from reactant to product. Each of these processes
has with it an associated energy barrier, and the slowest of these
processes is known as the rate determining step. If a large
energy barrier or overstep is required to be overcome for
nucleation, the possibility of metastable reactions occurring instead
of stable ones is accessed.
The current state of knowledge concerning reaction kinetics was discussed in Chapter 2 with a view to seeing what potential advances can be made in this study and what methods are available to obtain them. In this study, it has been decided to measure the systematic variation between porphyroblast size and heating rate observed in metamorphic rocks of known thermal and chemical history from the metamorphic aureole beneath the Rustenburg Layered Suite of the Bushveld Complex, South Africa to quantify the critical overstepping for nucleation, and an extensive suite of samples of andalusite-biotite-staurolite/cordierite bearing pelites showing this variation is examined. Central to this line of enquiry is the use of a textural parameter, b, a measure of the distribution of porphyroblast sizes in a population. Large porphyroblasts are observed to result from slow heating rates whilst faster heating gives rise to more hornfelsic textures. By using a forward model that simulates porphyroblastic nucleation and growth, the results of crystal size distribution (CSD) analysis for samples of known heating rate, when combined with a reasonable estimate for either the reaction rate or critical overstepping of reaction, allow the value of the other to be determined. Initial estimates for the overstepping have been obtained using a value for the forward reaction rate determined experimentally by Schramke et al, 1987, American Journal of Science, 287, 517-559.
The field area has been chosen because it acts as a 'natural laboratory' where the aureole rocks have been subjected to a single episode of metamorphism in the form of a thermal pulse without the complications of deformation. Field relationships show that the floor of the Rustenburg Layered Suite and the Pretoria Group metapelites dip concordantly at 15° in the Penge area to the north of Burgersfort. Further north, the dips rise to approximately 40°, whilst south of Burgersfort, dips are shallow and undulose. The tight constraints imposed by the geology in the Penge area allow precise models of emplacement to be constructed.
Chapter 4 presents a petrological discussion of the samples with the goal of defining a reaction sequence and relative timings of mineral growth which is compared with the predictions of equilibrium thermodynamic modelling. Pelitic lithologies are examined in terms of grade of metamorphism and in terms of Formation which allows similarities and differences to be highlighted. This is important since the intrusion gently cuts down into the stratigraphy in a northerly direction, exposing lower stratigraphic levels to higher grades of thermal metamorphism.
Geothermobarometric results are presented in Chapter 5 which allow constraints to be placed on the thickness of intrusion and peak metamorphic temperatures in the outer aureole, both of which have implications for the degree of lateral thinning of the Rustenburg Layered Suite which in turn affect the heating rates experienced by rocks of the outer aureole. Results were obtained from both conventional garnet-biotite thermometry and the use of Thermocalc in 'average PT' mode. It is seen in this chapter that samples from the outer part of the aureole suitable for textural analysis have reached peak metamorphic temperatures of 550 to 600° at approximately isobaric conditions of 3 kbar, whilst inner aureole temperatures reach up to 700°.
CSD and spatial analysis requires interpretation of a 2-D slice through a volume of rock. Chapter 6 considers the assumptions made in this process, for example the under-sampling of the small grains within a population and the distortion of true grain size created by taking a randomly orientated slice through a crystal. These effects combine to cancel one another out which means that for the larger size classes in a population the 2-D CSD is a good representative of the actual 3-D CSD. Methods of obtaining precise CSD results - tracing at scales ranging from thin-section to rock face, image digitization and a summary of the method of Cashman and Ferry, 1988, Contributions to Mineralogy and Petrology, 99, 401-415 - are presented. This method involves the fitting of a b parameter that describes the gradient of the log-linear portion of a plot of crystal size against population density, where such a straight line segment is present. If growth rates are assumed constant and linear, this kind of CSD shows the increase of nucleation rate with respect to time.
Chiastolite porphyroblasts in the outer aureole where rates of
heating have been slow show b
parameters of approximately 5 cm
The spatial distributions of the populations of crystals subject to CSD analysis do not show any significant departure towards ordering which would be expected if slow diffusion of nutrients required for crystal growth caused zones of depletion to form in which further nucleation was suppressed.
Field evidence, thermobarometric constraints and a postulated duration of intrusion of 75,000 years [Cawthorn and Walraven, 1998, Journal of Petrology, 39(9), 1669-1687] have allowed detailed modelling of the thermal pulse leaving the Rustenburg Layered Suite which is shown to have thinned in an easterly direction from 8 to 6 km. Heating rates have been quantified and vary from less than 0.1° per thousand years in outer aureole locations approximately 3 km stratigraphically below the contact to rates greater than 1° per thousand years in the highest grade locations that were suitable for CSD analysis approximately 1.5 km stratigraphically below the contact.
The CSD results obtained have been compared with those obtained using a numerical model [NuGrow] for the nucleation and growth of andalusite porphyroblasts [Waters, 1989, Terra Abstracts, 1, 297] which generate synthetic b parameters and oversteppings when rates of heating and reaction are supplied. Initial results in which simulations were performed used calculated heating rates, but assumed the forward rates of reaction experimentally determined by Schranke et al. (1987) which show that a temperature overstepping of between 5 and 10° is required to initiate andalusite nucleation. The estimated degree of overstepping is tied to the rate of forward reaction - higher oversteppings will be required if slower reaction rates are assumed.
The second half of Chapter 8 deals with an associated study [Waters, D. J. and Lovegrove, D. P., 2002. Assessing the extent of disequilibrium and overstepping of prograde metamorphic reactions in metapelites from the Bushveld Complex aureole, South Africa. Journal of Metamorphic Geology, 20, 135-149] in which the critical overstepping is evaluated using independent methods with the same suite of samples. Detailed microtextural study of graphite poor andalusite-staurolite-biotite hornfelses from the Upper Timeball Hill horizon 2.8 km stratigraphically below the base of the Rustenburg Layered Suite shows that the observed sequence of reaction is different to that predicted by equilibrium thermodynamic modelling. Chloritoid persists far beyond its predicted breakdown temperature to remain present into a period of simultaneous growth of andalusite, staurolite and biotite. The appearance (and subsequent disappearance) of cordierite during this interval is also inconsistent with the predictions of equilibrium thermodynamics and implies a delay in the nucleation of andalusite approaching 100° with respect to its predicted equilibrium temperature of formation. Replacement of early chloritoid crystals at constant volume by a range of chemically different minerals indicates mobility of chemical species through the intergranular medium, and that processes on the mineral surfaces themselves are rate limiting.
The predicted reactions for the formation of andalusite, staurolite and cordierite by chloritoid breakdown all have low entropy change of reaction. Formation of these minerals occurs once these lower entropy reactions have been overtaken in terms of reaction affinity by reactions predicted to be metastable with significantly higher entropy change, whence nucleation may begin. Such affinity calculations imply that the nucleation of andalusite requires a critical overstepping of over 4 kJ/mol porphyroblast formed, a value which translates to a 40° overstepping of the equilibrium temperature of the chlorite breakdown reaction. On a per gram atom oxygen basis, andalusite requires about 0.8 kJ for nucleation, whilst staurolite and cordierite require significantly lower oversteps of about 0.4 and 0.2 kJ respectively.
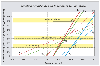
|
Diagram showing the variation of reaction affinity as a function of temperature which allows the evaluation of the critical overstepping for nucleation of andalusite, staurolite and cordierite. |
Differences between the amount of overstepping estimated by these two independent approaches may be explained by large differences between reaction rates determined experimentally and those operating in natural metamorphic environments. The coupling of nucleation rate, crystal growth rate and grain size distributions suggests that reactions occur at rates at least an order of magnitude slower than those measured in the laboratory by Schramke et al, 1987, and are comparable with those determined from Sr isotopic analysis of metamorphic terranes by Baxter and De Paulo, 2000, Science, 1411-1414.